Why are Diels Alder cycloadditions faster than uncatalyzed analogues?
It is well known that Diels–Alder reactions are greatly accelerated by Lewis acids (LAs) via complexation to the dienophile. 5 These LA-catalyzed cycloadditions are not only faster than their uncatalyzed analogues, but are also generally more regio- and stereoselective.
Is Diels Alder resea reversible?
The Diels–Alder (DA) reaction is regarded as quite a useful strategy in organic and macromolecular syntheses. The reversibility of this reaction and the advent of self-repair technology, as well as other applications in controlled macromolecular architectures and crosslinking, have strongly boosted the resea
What is Diels Alder chemistry?
Diels–Alder chemistry The DA reaction is a [4 + 2] cycloaddition of a conjugated diene and a substituted alkene (also termed dienophile) during which six π-electrons rearrange to form a cyclic, six-membered product ( Fig. 1 A).
Does Lewis acid assist Diels Alder reaction with Regio- and stereoselectivity?
Chen, Q., Chen, H., Meng, X. & Ma, Y. Lewis acid assisted Diels–Alder reaction with regio- and stereoselectivity: anti -1,4-Adducts with rigid scaffolds and their application in explosives sensing. Org. Lett. 17, 5016–5019 (2015).

Diels-Alder reaction Organic chemistry Khan Academy

16.5 Diels-Alder Reactions Organic Chemistry

Diels Alder Reaction
|
Catalytic Enantioselective Diels Alder Reaction: Application in the
25 jan. 2022 dedicated a chapter of a book to the cycloaddition reactions used for the synthesis of antiviral compounds [7] and a recent review described ... |
|
Trends in Diels Alder in Polymer Chemistry
5 oct. 2021 ical Society Reviews Royal Society of Chemistry |
|
Diels–Alder Reaction (Part 2)
The Diels-Alder Reaction. OT. Lecture Notes. Key Reviews: Total Synthesis. K.C. Nicolaou S.A. Snyder |
|
Inverse-Electron-Demand Diels-Alder Reactions of 2-Pyrones
25 nov. 2020 This reaction has been used for the synthesis of many biologically active natural products and drug candidates. In this review the developments ... |
|
Diels–Alder Cycloaddition Reactions in Sustainable Media
15 fév. 2022 In this review the most recent advances in the Diels–Alder reaction in environmentally benign and sustainable solvent systems since 2010 ... |
|
The Diels-Alder Reaction: an Update Introduction
Review. The Diels-Alder Reaction: an Update. Timothy John Brocksom Joanita Nakamura |
|
The Diels–Alder Reaction in Steroid Synthesis
17 nov. 2014 Abstract This review explores the myriad ways in which the Diels–. Alder reaction has been employed in the construction of the tetracyclic. |
|
The First General Enantioselective Catalytic Diels-Alder Reaction
(2) For recent reviews of enantioselective Diels-Alder reactions see: (a). Evans |
|
Metal Promoted Diels-Alder Reactions
31 jan. 2022 promoted Diels-Alder reactions are reviewed. Keywords: Diels-Alder reaction Catalysis |
|
Chitosan Hydrogels Based on the Diels–Alder Click Reaction
16 mar. 2022 It is reasonable therefore |
|
The Diels-Alder reaction
Diels-Alder (DA) reaction is incredibly valuable method for the synthesis of 6- rings • It is not within the If you are a little rusty on the Diels-Alder reaction either re-read your lecture notes or Summary of methods for stereoselective synthesis |
|
The First General Enantioselective Catalytic Diels-Alder Reaction
(2) For recent reviews of enantioselective Diels-Alder reactions, see: (a) Evans, D A ; Johnson, J S In ComprehensiVe Asymmetric Catalysis; Jacobsen, E N , |
|
The Diels-Alder Reaction: an Update Introduction
The Diels-Alder reaction is a pericyclic reaction under complete stereoelectronic control, but is strongly influenced by electronic and steric effects in both diene and dienophile However, the importance of these two effects is quite different in the intermolecular version and in the IMDA and TADA reactions |
|
Asymmetric DielsAlder Reactions
Asymmetric DielsAlder Reactions Myers Reviews: Corey, E J Angew Chem Int Ed 2002, 41, 1650–1667 Evans, D A ; Johnson, J S In Comprehensive |
|
Diels-Alder reactions in synthesis and method - UiO - DUO
The Diels-Alder reaction in its simplest form Both the diene and the dienophile can be heavily substituted16 The mechanism has been investigated extensively |
|
1 Asymmetric Catalysis of Diels–Alder Reaction - Wiley-VCH
synthesis of natural products and biologically important compounds The catalytic asymmetric aza Diels–Alder reaction is comprehensively reviewed in the |
|
The Diels-Alder reaction and biopolymer catalysis
Review The Diels-Alder reaction and biopolymer catalysis T M Tarasow and B E Eaton* Chemistry Division, NeXstar Pharmaceuticals, Inc , 2860 Wilderness |
|
[PDF] The Diels-Alder Reaction - MIT
Oct 5, 2007 · Regiochemical Course of the Diels Alder Reaction The "Ortho Para Rule" + + not not Review of quinolizidine and indolizidiene natural |
|
[PDF] 1 Asymmetric Catalysis of Diels–Alder Reaction - Wiley-VCH
The Diels–Alder (DA) reaction (or “diene synthesis”) between a diene and a dienophile generates two s bonds stereoselectively and up to four chiral centers in a single step to afford six membered cyclic compounds For their discovery, Diels and Alder received the Nobel Prize in chemistry in 1950 |
|
The Diels-Alder reaction and biopolymer catalysis
Review The Diels Alder reaction and biopolymer catalysis T M Tarasow and B E Eaton* Chemistry Division, NeXstar Pharmaceuticals, Inc, 2860 Wilderness |
|
[PDF] Asymmetric DielsAlder Reactions
Asymmetric DielsAlder Reactions Myers Reviews Corey, E J Angew Chem Int Ed 2002, 41, 1650–1667 Evans, D A; Johnson, J S In Comprehensive |
- Diels-Alder reaction examples
- diels-alder reaction practice problems
- Diels-Alder reaction mechanism PDF
- Diels-Alder reaction practice pdf
- Diels-Alder reaction ppt
- Application of Diels-Alder reaction
- Diels-Alder adduct
- Predicting Diels-Alder products

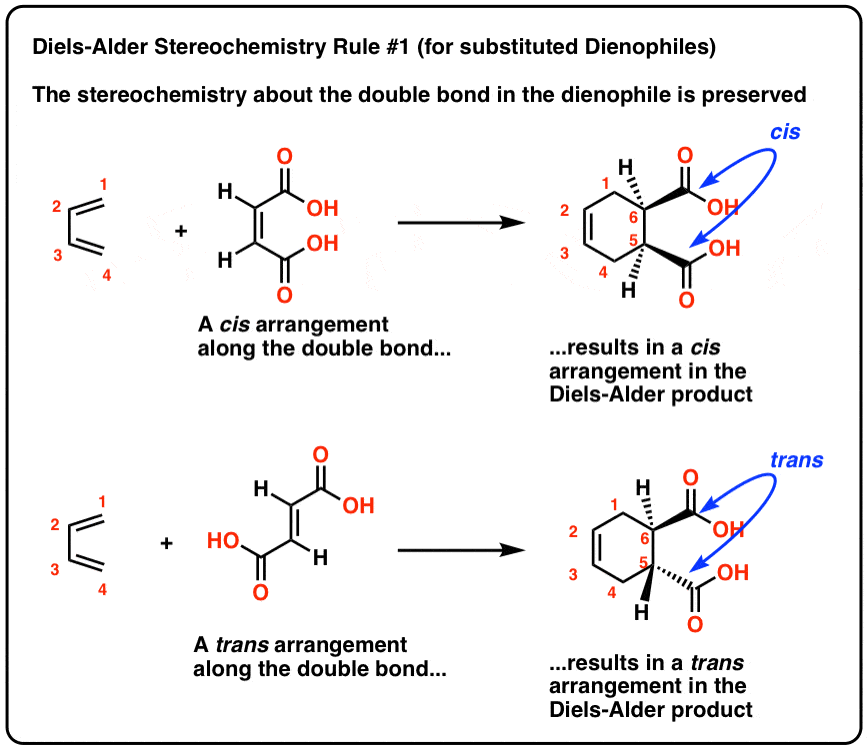
Stereochemistry of the Diels-Alder Reaction – Master Organic Chemistry
Source:https://0.academia-photos.com/attachment_thumbnails/34282011/mini_magick20180818-12558-1f1iugb.png?1534589714
PDF) Introduction To Diels Alder Reaction Its Mechanism And
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/3-diels-alder-stereochemistry-rule-1-for-substituted-dienophlies-stereochemistry-about-double-bond-in-dienophile-is-preserved-.gif

Stereochemistry of the Diels-Alder Reaction – Master Organic Chemistry
Source:https://i1.rgstatic.net/publication/323062184_Diels-Alder_Reaction_of_IsobenzofuransCyclopentadienones_with_Tetrathiafulvalene_Preparation_of_Naphthalene_Fluoranthene_and_Fluorenone_Derivatives/links/5c0a3a42a6fdcc494fdfd807/largepreview.png
PDF) Diels–Alder Reaction of Isobenzofurans/Cyclopentadienones
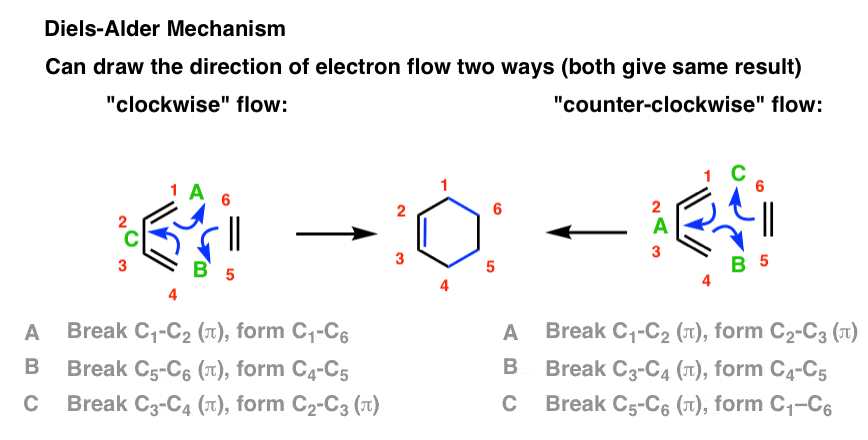
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/4-arrow-pushing-mechanism-for-diels-alder-reaction-can-be-drawn-two-ways-either-with-clockwise-or-counter-clockwise-flow-of-electrons.gif

The Diels-Alder Reaction – Master Organic Chemistry
Source:https://www.eurekaselect.com/images/graphical-abstract/cos/15/1/004.jpg

Hetero-Diels-Alder Reactions in the Synthesis of Biologically
Source:https://www.tsijournals.com/articles-images/organic-chemistry-14-3-131-g008.png
diels alder reaction
[PDF] Diels-Alder Reaction:
- Diels-Alder reaction Practice Problems pdf
- Diels-Alder reaction PDF
- Diels-Alder reaction examples
- Introduction of Diels-Alder reaction
- Diels–Alder reactions in water - iupaciupac.org › publications › pac
- significant additional benefits when it is used as a solvent for the Diels–Alder reaction. The mechanism by which the unique properties of water enhance the rate ...[PDF] The Type II Intramolecular Diels-Alder Reaction: Synthesis and ...gbdong.cm.utexas.edu › seminar › old › The Type II Intramolecular ...
- The Type II Intramolecular Diels-Alder Reaction: Synthesis and Chemistry of Bridgehead Alkenes. 2013. 10. 24. Haye Min Ko ...[PDF] Thermodynamic and Kinetic Study of Diels–Alder Reaction ... - MDPIwww.mdpi.com › pdf
- Jan 7
- 2020 · Endo and exo isomers were distinguished in 1H NMR
- and the transition from a kinetic
- controlled Diels–Alder reaction to a thermodynamic one ...[PDF] Diels-Alder Reaction:www.linfield.edu › assets › files › chem › Courses › diels_alder
- CHEM 322: Diels-Alder Reaction: Preparation of cis-norbornene-2
- 3-endo- dicarboxylic anhydride. Introduction: In 1921
- Otto Diels and Kurt Alder discovered ...Related searchesAsymmetric Diels--Alder reaction PDF
- Diels-Alder reaction purpose
- Intramolecular Diels-Alder reaction PDF
- Enantioselective Diels--Alder reaction
- Diels-Alder reaction with alkyne
- Does the Diels-Alder reaction have stereoselectivity
- Diels-Alder reaction balanced equation
- Diels-Alder reaction procedure
diels alder reaction balanced equation
[PDF] Diels-Alder Reaction
- Diels-Alder reaction examples
- Maleic anhydride
- Diels-Alder reaction PDF
- 3-sulfolene and maleic anhydride reaction
- [PDF] Diels-Alder reaction between naphthalene and N ... - Arkivocwww.arkat-usa.org › get-file
- increases the rate of the Diels-Alder reaction of dienes.2-4 Benzene does not ... interatomic C(1)-C(4) distance in the diene (R1-4)
- and balance of energies of ... equilibrium constant of the π
- π-complex between reactants 2 and 7(equation 2).Kinetics of Isoprene-Maleic Anhydride Diels-Alder Reaction in ...www.jstage.jst.go.jp › article › apcche › _pdf
- to examine the reaction mechanism in the vicinity of the critical point. The following Diels-Alder reaction of isoprene with maleic anhydride in supercritical carbon ...[PDF] Diels-Alder PDFwww.bc.edu › mcas › pdf › undergrad › lab-courses › org › spring
- The simplest Diels-Alder reaction is between 1
- 3-butadiene and ethylene: +. The mechanism of the reaction begins with the diene assuming the higher energy
- .[PDF] Diels-Alder Reactionwww.cerritos.edu › chemistry › chem_212 › Documents › Lab › 2_D...
- Figure 1: The overall equation. The Diels-Alder reaction is a member of a larger class of pericyclic reactions. These are reactions with transition states in which the ...Related searchesDiscuss the applications of Diels-Alder reaction
- Anthracene undergoes Diels-Alder reaction
- Petroleum ether in Diels-Alder reaction
- anthracene-maleic anhydride diels-alder adduct melting point
- 1 3-butadiene maleic anhydride mechanism
- what role does 3-sulfolene play in the diels-alder reaction
- Why is xylenes used as the reflux solvent for the Diels-Alder experiment
- Diels-Alder reaction notes
diels alder reaction examples
[PDF] Diels Alder Stereochemistry Worksheet This is not an assignment
- Diels-Alder reaction PDF
- Diels-Alder reaction practice pdf
- Diels-Alder reaction practice problems
- Diels-Alder reaction ppt
- [PDF] Diels – Alder Reaction 1
- 4-CycloAddition Reaction of Dienesww2.chemistry.gatech.edu › class › Chem-2312-August-27-handout
- What's Going With Respect to a Diels-Alder 1
- 4 Cycloaddition Reaction? X. X. +. Diene. S-cis ... Examples of Dienes and Dienophiles. Prefer electron ...The intramolecular Diels–Alder reaction - Canadian Science ...www.nrcresearchpress.com › doi › pdfplus
- Chem. 62
- 183 (1984). Recent advances and examples of the intramolecular Diels-Alder reaction are summarized and applications to the total synthesis ...Theoretical exammation of the Diels–Alder reaction of 1
- 3-butadiene ...www.nrcresearchpress.com › doi › pdfplus
- Abstract: All Diels-Alder reactions between 1
- 3-butadiene and cyclopentadiene or 2H-phosphole ... have been carried out; however
- there are limited examples.[PDF] The Diels-Alder Reaction in Total Synthesis - Moodle@Unitsmoodle2.units.it › content › 11_ACIE_Nicolau_Diels_Alder
- natural product synthesis
- reaction mechanism and design
- and application of these fields to chemical ... this example is that the use of SnCl4 in the Diels±Alder .[PDF] Diels Alder Stereochemistry Worksheet This is not an assignment ...www.reed.edu › chemistry › alan › Models05 › DAstereo_worksheet
- This worksheet provides some examples of Diels-Alder reactions and ... Diels- Alder reaction normally makes a cyclohexene product
- but it makes a bicyclic ring .Related searchesDiels-Alder adduct
- Diels-Alder reaction stereochemistry
- Predicting Diels-Alder products
- Diels-Alder reaction balanced equation
- Does the Diels-Alder reaction have stereoselectivity
- Diels-Alder practice Quiz
- Diels-Alder reaction procedure
- Reactivity of dienes in Diels-Alder
diels alder reaction mechanism pdf
[PDF] A Diels Alder reaction - Semantic Scholar
- Diels-Alder reaction practice pdf
- Diels-Alder reaction practice problems
- Diels-Alder reaction ppt
- Diels-Alder reaction examples
- [PDF] CYCLOADDITIONS IN ORGANIC SYNTHESISprofiles.uonbi.ac.ke › andakala › files › sch_504_cycloadditions_in_o...
- Although the Diels Alder reaction combines a diene and a dienophile to generate a cyclohexene
- the reaction between simple 1
- 3- butadiene and ethene is very ...[PDF] Diels – Alder Reaction 1
- 4-CycloAddition Reaction of Dienesww2.chemistry.gatech.edu › class › Chem-2312-August-27-handout
- 4. Examples of Dienes and Dienophiles. Prefer electron withdrawing groups on dienophile. Prefer electron donating groups on diene ...[PDF] Electrophilic Addition Reactions of Dienes 1. Mechanism - Chemistrychemistry.bd.psu.edu › justik › CHEM 212 CH 13 Diels-Alder
- The Diels-‐Alder reaction is a one-‐step concerted process between a diene and dienophile to afford a cyclohexne: 2. Since two new sigma bonds are formed with ...[PDF] The Diels±Alder Reactionwww.mobt3ath.com › uplode › book › book-36001
- enzyme has provided insights into the mechanism of biosynthetic cycloaddition. As a follow-up to our book Dienes in the Diels±Alder Reaction (1990) and in.[PDF] The Diels-Alder Reaction - MITweb.mit.edu › www
- Oct 5
- 2007 · construction
- it was clear by 1970 that the Diels-Alder reaction would be ... Suprafacial with respect to the Dienophile Component. H. EWG. RE.[PDF] 34 - MSU chemistrywww2.chemistry.msu.edu › courses › cem850 › F12_WULFF
- Solutions Manual to accompany Organic Chemistry 2e. MeO2C. H. OMe. Me3SiO . H ... So electronic factors dominate
- perhaps because the dienophile has.[PDF] A Diels Alder reaction - Semantic Scholarpdfs.semanticscholar.org › ...
- relative orientations could either be endo or exo. DA reaction of cyclopentadien analogue. (furan) and the specified dienophile (succinimide) is shown below. O.Related searchesDiels-Alder reaction stereochemistry
- Predicting Diels-Alder products
- Reactivity of dienes in Diels-Alder
- Diels-Alder adduct
- Diels-Alder reaction balanced equation
- Diels-Alder: regiochemistry
- Does the Diels-Alder reaction have stereoselectivity
- Diels − Alder